By Jerry Underwood
For The Birmingham Times
Scientists at Birmingham-based Southern Research are front-runners in the global race to develop new vaccines and therapeutic treatments against COVID-19, the disease caused by the novel coronavirus that is blamed for a growing death toll and massive economic fallout worldwide.
The work now taking place in the organization’s laboratories builds upon decades of multi-faceted research in infectious diseases and efforts targeting discovery and development of medicines to treat numerous illnesses.
“We fully understand the urgency and need to accelerate drug discovery and development activities to help curb the coronavirus pandemic,” said Michael Catalano, Southern Research’s interim chief operating officer. “Southern Research is honored to play a role in the world’s health and safety. Biological science developments, such as this collaboration, are core to our nearly 80-year mission.”
Founded in 1941, Southern Research is a nonprofit, internationally-known scientific research organization with more than 400 scientists and engineers working across four divisions: Drug Discovery, Drug Development, Engineering, and Energy & Environment. It is headquartered in Birmingham with additional laboratories and offices in Wilsonville, Alabama; Frederick, Maryland; and Houston, Texas.
Southern Research, which provides commercial and government clients with nonclinical and clinical trial support services, is headquartered at 2000 9th Avenue South which houses its main offices, its drug discovery and its development divisions.
The company is one of Birmingham’s largest non-profits with projects that range from developing new medical devices, launching manned missions to Mars, and working on behalf of entities like the National Cancer Institute, the U.S. Department of Defense, major aerospace firms and other private and government organizations.
Since SARS-CoV-2 emerged as a global threat earlier this year, Southern Research scientists have been engaged in multiple fronts in the battle against the virus that causes COVID-19.
In February, Southern Research announced a research collaboration with Tonix Pharmaceuticals Holding Co. to support development of a COVID-19 vaccine candidate being advanced by the New York-based clinical-stage biopharmaceutical firm.
Southern Research and Tonix are testing a potential vaccine that expresses a protein from SARS-CoV-2 as a means of offering protection against the disease.
The scientists at Southern Research were of the first to have developed capabilities for evaluating potential new therapies against COVID-19. Towards this accomplishment, they have been working with several major pharmaceutical companies, biotech companies, the Bill & Melinda Gates Foundation and The National Center for Advancing Translational Sciences, an agency within the National Institutes of Health (NIH).
The organization’s scientists are credited with the discovery of seven Food and Drug Administration (FDA) approved anti-cancer drugs.
“As part of our long history aiming to identify new medicines to treat life-threatening diseases, such as cancer and HIV, we were well positioned to quickly address the COVID-19 pandemic,” said Mark J. Suto, Ph.D., vice president of Southern Research’s Drug Discovery division and interim vice president of its Drug Development Division.
Southern Research was founded in Birmingham in October 1941 as the Alabama Research Institute. In 2015, it dropped the “institute” to become simply Southern Research.
INTERNATIONALLY KNOWN
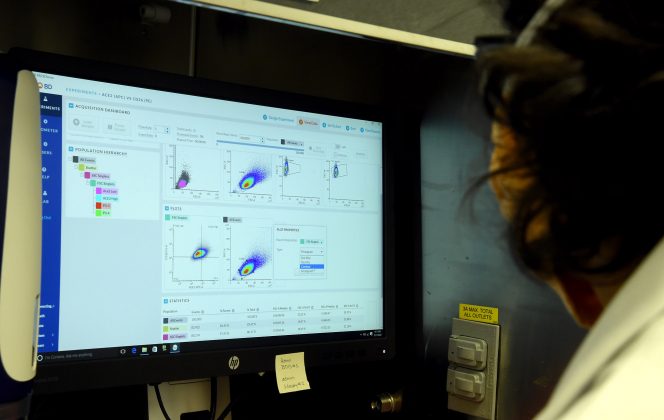
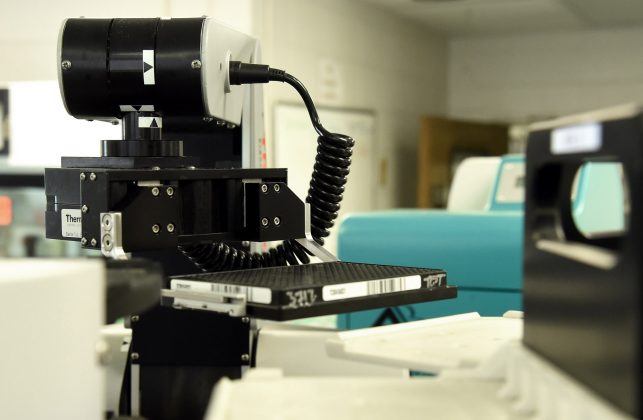
A key asset is Southern Research’s state-of-the-art High-Throughput Screening (HTS) Center, which features advanced robotic equipment, and can be used to test new compounds and carefully manage the immense amount of resulting data. This system is an essential capability that is especially valuable in drug discovery.
Southern Research’s HTS Center, which is internationally known for its work infectious diseases, can conduct these studies under the required containment needed for highly infectious agents such as SARS-CoV-2.
Over the years, the HTS Center has screened more than 15 million compound samples in over 50 different infectious disease assays. It currently serves as the screening core for the NIH-funded Antiviral Drug Discovery and Development Center (AD3C), based at the University of Alabama at Birmingham (UAB), where there are several close collaborators on many projects. The group has also collaborated with the Division of AIDS, which is part of the National Institute Allergy and Infectious Diseases, as well as foundations such as the Cystic Fibrosis Foundation.
“The goal is to discover drugs that can be used as therapeutic treatment for COVID-19. By screening compounds in our HTS Center, we can rapidly identify those that exhibit antiviral effects against SARS-CoV-2,” said Robert Bostwick, Ph.D., director of the HTS Center.
“These compounds can then be used as starting points for the development of new therapeutic agents,” he added.
‘DRUG REPURPOSING’
The HTS Center is an important part of an internal research program launched by Southern Research that focuses on COVID-19. The goal is to identify drugs already approved by the FDA for other uses that may be effective against SARS-CoV-2.
“In the case of combating COVID-19, speed is of the essence given wide-reaching consequences. It is well-known that the development of new drugs is a costly endeavor and requires years of research,” said Suto, who has made wide-ranging contributions to pharmaceutical research during a career spanning more than 35 years.
“Southern Research has taken an approach which could result in the identification of new therapies in a period of months rather than years,” Suto said.
Already, Southern Research has tested thousands of drugs and found several that are highly active against the virus, the majority of which were not developed as antivirals in the first place.
“It takes years to bring a new drug to market. Since the safety profile of marketed drugs is already known, the path to clinical studies to determine if it can effectively treat a disease other than the one for which it is marketed is much faster,” Bostwick said.
“Therefore, in a pandemic it is much quicker to repurpose existing drugs for use in combating the pathogen as opposed to inventing an entirely new drug,” Bostwick added.
The internal program is being funded by the COVID-19 Research Acceleration Fund, which is supported by generous donors and friends of Southern Research. It is anticipated that any human phase trials stemming from the program will be conducted at UAB.
In addition, Southern Research and UAB are working with California-based Scripps Research to screen Scripps’ repurposed library of compounds to determine if any licensed drugs have activity against SARS-CoV-2.
EXPANDING RESEARCH
Meanwhile, scientists at Southern Research continue to explore new avenues in the effort to combat COVID-19. In July, the organization announced an expansion of its strategic collaboration with Tonix Pharmaceuticals to include a study of immune system responses to SARS-CoV-2.
This research will focus specifically on T cell immune responses to SARS-CoV-2 in volunteers who have recovered from the disease or who remain asymptomatic after exposure to COVID-19.
Raj Kalkeri, Ph.D., MBA, a senior scientist in Southern Research’s Infectious Disease Research Group, said the study’s findings will shed new light on the role that T cells – a central part of the immune system – may play in the development of a vaccine against the novel coronavirus.
“As scientists, we know that the most successful vaccines mimic and potentiate how the immune system responds to insult such as a virus,” Kalkeri said. “This additional work we are doing with Tonix will add focus to that objective.”
Catalano, the interim COO at Southern Research, said he is confident that the organization’s scientists will continue to make important contributions in the global effort to overcome a disease that has killed more than 160,000 Americans.
“Despite the numerous strides Southern Research has undertaken to provide novel therapeutics against COVID-19, a great deal of work remains to accelerate identification of therapeutics against the disease, allowing for clinical trials to be initiated rapidly,” Catalano said.
“Work performed now will also help improve responses in the future.”
RESEARCH HERITAGE
Southern Research’s broad efforts against coronavirus directly align with the organization’s historical mission to discover and development therapeutics used in treating diseases. After the organization initiated a cancer research program in 1946, it made pioneering breakthroughs in chemotherapy techniques and discovered seven FDA-approved drugs that have been used in cancer treatment.
Moreover, Southern Research maintains a long-standing history with the National Cancer Institute (NCI), including its role in the NCI Experimental Therapeutics (NExT)’s Chemical Biology Consortium.
Southern Research has long been involved in research on infectious diseases, including HIV/AIDS. It joined the front lines in the battle against HIV/AIDS in 1986, not long after the deadly viral infection emerged in the U.S. as a new public health threat. Since then, it has helped 90 percent of all currently approved antivirals used against the disease get to market.
Southern Research also has been heavily involved in U.S. government influenza programs since 2004, when H5N1, or bird flu, emerged as a serious threat. Since 2009, the organization has supported the government as a primary provider of flu vaccine testing and support.
In addition, Southern Research scientists have conducted research into mosquito-borne viruses such as Zika, chikungunya, dengue and yellow fever.
Editor’s Note: Jerry Underwood has done public relations for Southern Research and some of this material originally appeared on Southern Research’s website. This story was assigned as part of a broader package of articles on entities and scientists working on COVID-19 treatments.
Meet the minority researchers focused on a coronavirus treatment
The need for vaccine trials to reflect America’s diversity
Glenn Ellis: Why some blacks may be skeptical of vaccine treatments